What Is the Ph When 1.5 Ml of 3.0 M Hcl Is Added to 1.0 L of the Buffer?
Chapter 14. Acid-Base Equilibria
14.half-dozen Buffers
Learning Objectives
By the end of this section, you will be able to:
- Describe the composition and function of acid–base buffers
- Calculate the pH of a buffer before and after the addition of added acrid or base of operations
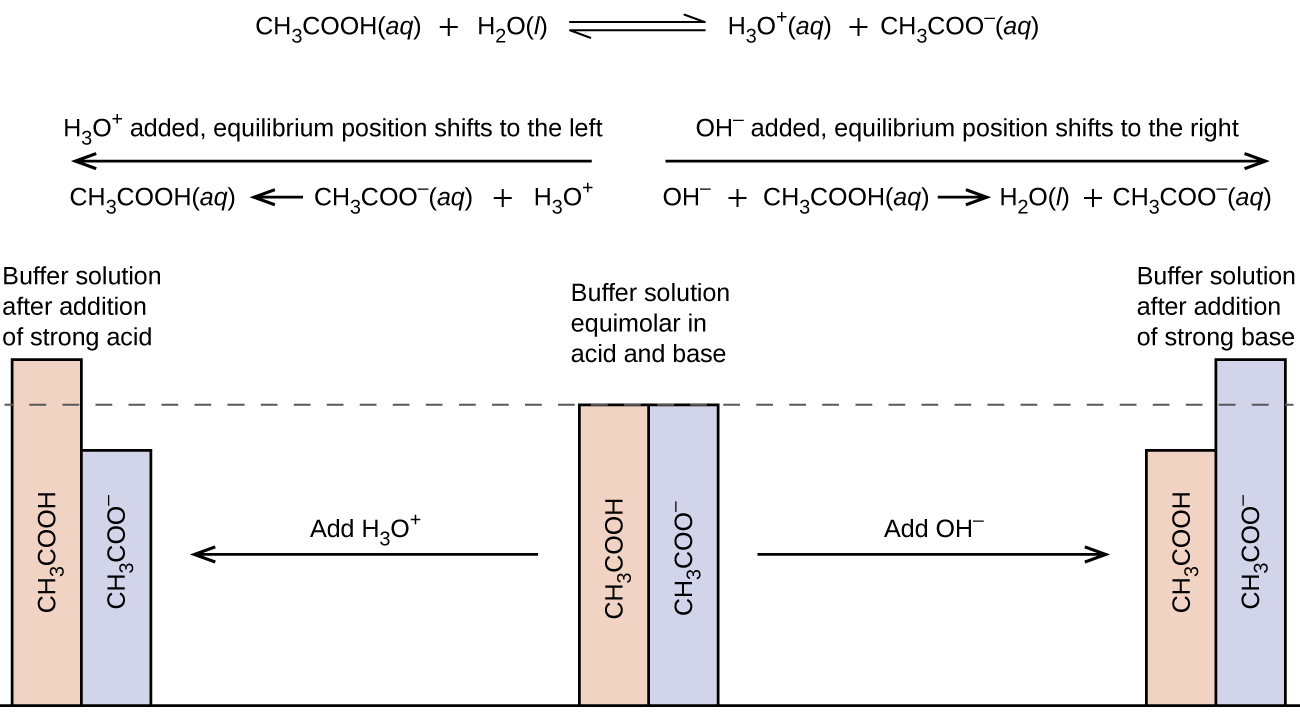
A mixture of a weak acrid and its conjugate base (or a mixture of a weak base and its conjugate acrid) is chosen a buffer solution, or a buffer. Buffer solutions resist a change in pH when pocket-sized amounts of a strong acid or a strong base of operations are added (Figure 1). A solution of acerb acid and sodium acetate (CHiiiCOOH + CHiiiCOONa) is an example of a buffer that consists of a weak acid and its common salt. An instance of a buffer that consists of a weak base of operations and its common salt is a solution of ammonia and ammonium chloride (NH3(aq) + NH4Cl(aq)).

How Buffers Work
A mixture of acetic acrid and sodium acetate is acidic because the Thou a of acetic acid is greater than the M b of its conjugate base acetate. It is a buffer because it contains both the weak acid and its salt. Hence, it acts to go on the hydronium ion concentration (and the pH) near constant by the improver of either a modest corporeality of a strong acid or a stiff base. If we add a base of operations such as sodium hydroxide, the hydroxide ions react with the few hydronium ions nowadays. Then more than of the acerb acid reacts with water, restoring the hydronium ion concentration almost to its original value:
[latex]\text{CH}_3\text{CO}_2\text{H}(aq)\;+\;\text{H}_2\text{O}(l)\;{\longrightarrow}\;\text{H}_3\text{O}^{+}(aq)\;+\;\text{CH}_3\text{CO}_2^{\;\;-}(aq)[/latex]
The pH changes very little. If we add an acid such as muriatic acid, most of the hydronium ions from the hydrochloric acid combine with acetate ions, forming acetic acid molecules:
[latex]\text{H}_3\text{O}^{+}(aq)\;+\;\text{CH}_3\text{CO}_2^{\;\;-}(aq)\;{\longrightarrow}\;\text{CH}_3\text{CO}_2\text{H}(aq)\;+\;\text{H}_2\text{O}(fifty)[/latex]
Thus, there is very little increase in the concentration of the hydronium ion, and the pH remains practically unchanged (Effigy 2).
A mixture of ammonia and ammonium chloride is bones because the K b for ammonia is greater than the K a for the ammonium ion. It is a buffer because it also contains the salt of the weak base. If we add a base (hydroxide ions), ammonium ions in the buffer react with the hydroxide ions to form ammonia and water and reduce the hydroxide ion concentration almost to its original value:
[latex]\text{NH}_4^{\;\;+}(aq)\;+\;\text{OH}^{-}(aq)\;{\longrightarrow}\;\text{NH}_3(aq)\;+\;\text{H}_2\text{O}(l)[/latex]
If nosotros add an acid (hydronium ions), ammonia molecules in the buffer mixture react with the hydronium ions to grade ammonium ions and reduce the hydronium ion concentration almost to its original value:
[latex]\text{H}_3\text{O}^{+}(aq)\;+\;\text{NH}_3(aq)\;{\longrightarrow}\;\text{NH}_4^{\;\;+}(aq)\;+\;\text{H}_2\text{O}(l)[/latex]
The iii parts of the post-obit case illustrate the change in pH that accompanies the addition of base to a buffered solution of a weak acrid and to an unbuffered solution of a potent acid.
Example 1
pH Changes in Buffered and Unbuffered Solutions
Acetate buffers are used in biochemical studies of enzymes and other chemical components of cells to prevent pH changes that might change the biochemical activity of these compounds.
(a) Summate the pH of an acetate buffer that is a mixture with 0.x 1000 acetic acrid and 0.ten 1000 sodium acetate.
Solution
To decide the pH of the buffer solution nosotros utilise a typical equilibrium adding (every bit illustrated in before Examples):
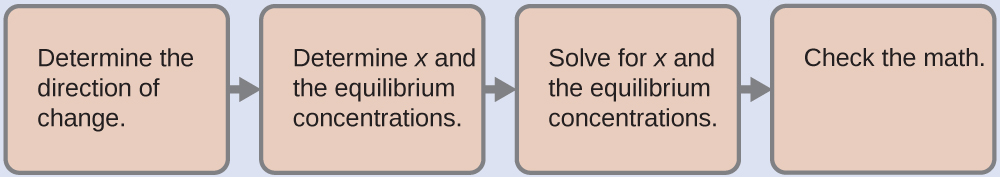
- Determine the direction of alter. The equilibrium in a mixture of H3O+, [latex]\text{CH}_3\text{CO}_2^{\;\;-}[/latex], and CH3CO2H is:
[latex]\text{CH}_3\text{CO}_2\text{H}(aq)\;+\;\text{H}_2\text{O}(l)\;{\rightleftharpoons}\;\text{H}_3\text{O}^{+}(aq)\;+\;\text{CH}_3\text{CO}_2^{\;\;-}(aq)[/latex]
The equilibrium constant for CH3COiiH is not given, so we await it up in Appendix H: K a = ane.eight × ten−5. With [CH3CO2H] = [latex][\text{CH}_3\text{CO}_2^{\;\;-}][/latex] = 0.10 M and [HthreeO+] = ~0 One thousand, the reaction shifts to the correct to course H3O+.
- Make up one's mind x and equilibrium concentrations. A table of changes and concentrations follows:
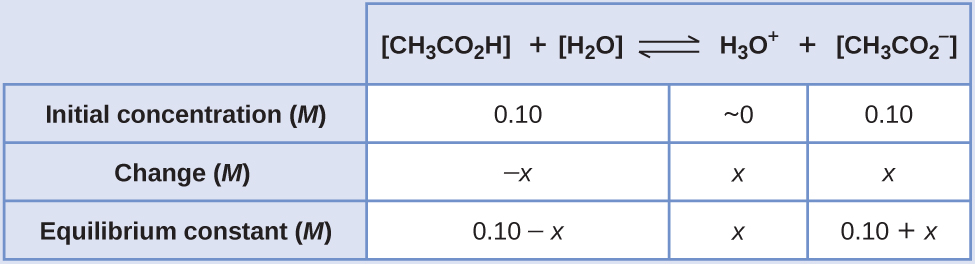
- Solve for x and the equilibrium concentrations. We find:
[latex]ten = 1.viii\;\times\;10^{-5}\;M[/latex]
and
[latex][\text{H}_3\text{O}^{+}] = 0\;+\;x = i.eight\;\times\;10^{-v}\;G[/latex]
Thus:
[latex]\text{pH} = -\text{log}[\text{H}_3\text{O}^{+}] = -\text{log}(1.viii\;\times\;x^{-5})[/latex]
[latex]= iv.74[/latex]
- Cheque the piece of work. If we calculate all calculated equilibrium concentrations, we find that the equilibrium value of the reaction coefficient, Q = K a.
(b) Summate the pH after one.0 mL of 0.10 M NaOH is added to 100 mL of this buffer, giving a solution with a volume of 101 mL.
First, we calculate the concentrations of an intermediate mixture resulting from the complete reaction between the acid in the buffer and the added base. Then we determine the concentrations of the mixture at the new equilibrium:
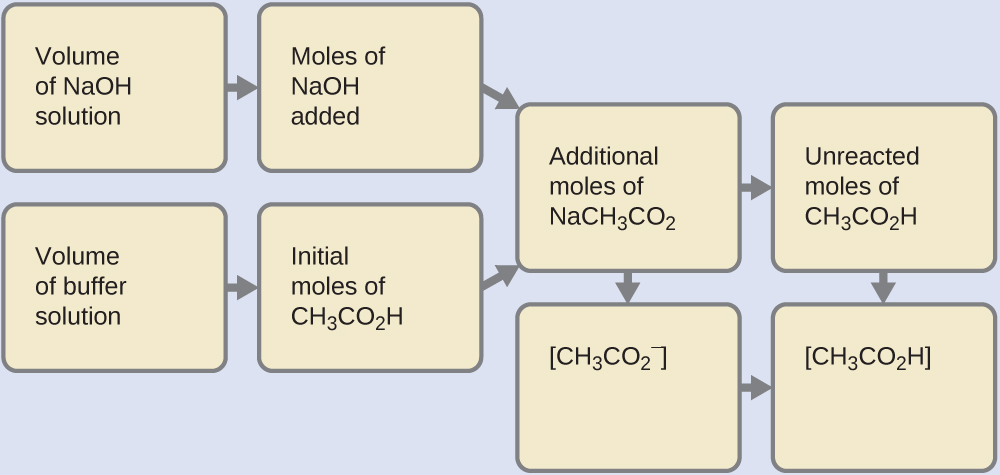
- Determine the moles of NaOH. 1 milliliter (0.0010 L) of 0.10 M NaOH contains:
[latex]0.0010\;\dominion[0.75ex]{0.7em}{0.1ex}\hspace{-0.7em}\text{L}\;\times\;(\frac{0.ten\;\text{mol\;NaOH}}{ane\;\rule[0.375ex]{0.5em}{0.1ex}\hspace{-0.5em}\text{50}}) = 1.0\;\times\;x^{-4}\;\text{mol\;NaOH}[/latex]
- Determine the moles of CH2CO2H. Before reaction, 0.100 Fifty of the buffer solution contains:
[latex]0.100\;\rule[0.75ex]{0.7em}{0.1ex}\hspace{-0.7em}\text{Fifty}\;\times\;(\frac{0.100\;\text{mol\;CH}_3\text{CO}_2\text{H}}{1\;\dominion[0.375ex]{0.5em}{0.1ex}\hspace{-0.5em}\text{L}}) = i.00\;\times\;10^{-2}\;\text{mol\;CH}_3\text{CO}_2\text{H}[/latex]
- Solve for the amount of NaCH3COtwo produced. The one.0 × ten−4 mol of NaOH neutralizes 1.0 × ten−4 mol of CH3COiiH, leaving:
[latex](one.0\;\times\;10^{-2})\;-\;(0.01\;\times\;x^{-2}) = 0.99\;\times\;10^{-2}\;\text{mol\;CH}_3\text{CO}_2\text{H}[/latex]
and producing one.0 × 10−iv mol of NaCHiiiCO2. This makes a full of:
[latex](i.0\;\times\;10^{-ii})\;+\;(0.01\;\times\;x^{-2}) = one.01\;\times\;10^{-2}\;\text{mol\;NaCH}_3\text{CO}_2[/latex]
- Find the molarity of the products. Subsequently reaction, CHiiiCO2H and NaCHthreeCO2 are contained in 101 mL of the intermediate solution, so:
[latex][\text{CH}_3\text{CO}_2\text{H}] = \frac{nine.9\;\times\;x^{-3}\;\text{mol}}{0.101\;\text{L}} = 0.098\;Grand[/latex]
[latex][\text{NaCH}_3\text{CO}_2] = \frac{ane.01\;\times\;10^{-2}\;\text{mol}}{0.101\;\text{50}} = 0.100\;M[/latex]
Now we calculate the pH after the intermediate solution, which is 0.098 M in CH3COiiH and 0.100 Thou in NaCH3COii, comes to equilibrium. The adding is very similar to that in role (a) of this example:
This series of calculations gives a pH = 4.75. Thus the addition of the base barely changes the pH of the solution (Figure 1).
(c) For comparing, calculate the pH after 1.0 mL of 0.10 M NaOH is added to 100 mL of a solution of an unbuffered solution with a pH of four.74 (a 1.8 × 10−5–M solution of HCl). The volume of the final solution is 101 mL.
Solution
This i.viii × x−5–Yard solution of HCl has the same hydronium ion concentration every bit the 0.10-M solution of acerb acrid-sodium acetate buffer described in part (a) of this example. The solution contains:
[latex]0.100\;\text{L}\;\times\;(\frac{1.8\;\times\;10^{-five}\;\text{mol\;HCl}}{1\;\text{L}}) = 1.8\;\times\;ten^{-6}\;\text{mol\;HCl}[/latex]
Every bit shown in role (b), ane mL of 0.10 M NaOH contains 1.0 × 10−four mol of NaOH. When the NaOH and HCl solutions are mixed, the HCl is the limiting reagent in the reaction. All of the HCl reacts, and the amount of NaOH that remains is:
[latex](one.0\;\times\;10^{-iv})\;-\;(ane.8\;\times\;ten^{-6}) = 9.8\;\times\;10^{-v}\;G[/latex]
The concentration of NaOH is:
[latex]\frac{9.viii\;\times\;10^{-v}\;M\;\text{NaOH}}{0.101\;\text{L}} = 9.7\;\times\;ten^{-4}\;Thou[/latex]
The pOH of this solution is:
[latex]\text{pOH} = -\text{log}[\text{OH}^{-}] = -\text{log}(9.7\;\times\;10^{-4}) = 3.01[/latex]
The pH is:
[latex]\text{pH} = xiv.00\;-\;\text{pOH} = 10.99[/latex]
The pH changes from iv.74 to x.99 in this unbuffered solution. This compares to the change of 4.74 to 4.75 that occurred when the same amount of NaOH was added to the buffered solution described in function (b).
Check Your Learning
Show that adding 1.0 mL of 0.x G HCl changes the pH of 100 mL of a 1.8 × 10−5 M HCl solution from 4.74 to iii.00.
Answer:
Initial pH of 1.8 × 10−v M HCl; pH = −log[H3O+] = −log[1.8 × ten−five] = 4.74
Moles of H3O+ in 100 mL one.8 × 10−5 M HCl; i.viii × 10−5 moles/L × 0.100 L = i.8 × 10−6
Moles of H3O+ added by add-on of 1.0 mL of 0.10 K HCl: 0.10 moles/L × 0.0010 L = 1.0 × 10−4 moles; final pH after addition of 1.0 mL of 0.10 Thou HCl:
[latex]\text{pH} = -\text{log}[\text{H}_3\text{O}^{+}] = -\text{log}(\frac{\text{full\;moles\;H}_3\text{O}^{+}}{\text{total\;volume}}) = -\text{log}(\frac{1.0\;\times\;10^{-iv}\;\text{mol}\;+\;one.8\;\times\;10^{-vi}\;\text{mol}}{101\;\text{mL}(\frac{i\;\text{50}}{thou\;\text{mL}})}) = 3.00[/latex]
If we add together an acid or a base to a buffer that is a mixture of a weak base of operations and its salt, the calculations of the changes in pH are analogous to those for a buffer mixture of a weak acid and its salt.
Buffer Capacity
Buffer solutions practise not accept an unlimited capacity to go along the pH relatively constant (Effigy 3). If we add then much base to a buffer that the weak acid is wearied, no more buffering action toward the base of operations is possible. On the other paw, if we add an excess of acid, the weak base would be exhausted, and no more buffering action toward whatsoever boosted acid would be possible. In fact, we do not even need to exhaust all of the acid or base of operations in a buffer to overwhelm information technology; its buffering action will diminish rapidly as a given component nears depletion.

The buffer capacity is the amount of acrid or base of operations that tin can be added to a given volume of a buffer solution before the pH changes significantly, usually past one unit. Buffer capacity depends on the amounts of the weak acid and its conjugate base of operations that are in a buffer mixture. For example, ane L of a solution that is 1.0 1000 in acerb acrid and 1.0 M in sodium acetate has a greater buffer capacity than i L of a solution that is 0.10 G in acetic acid and 0.10 K in sodium acetate even though both solutions have the same pH. The first solution has more buffer chapters because it contains more acetic acid and acetate ion.
Choice of Suitable Buffer Mixtures
There are two useful rules of thumb for selecting buffer mixtures:
- A skillful buffer mixture should take about equal concentrations of both of its components. A buffer solution has by and large lost its usefulness when one component of the buffer pair is less than about 10% of the other. Figure 4 shows an acetic acrid-acetate ion buffer equally base of operations is added. The initial pH is 4.74. A change of one pH unit occurs when the acetic acid concentration is reduced to 11% of the acetate ion concentration.
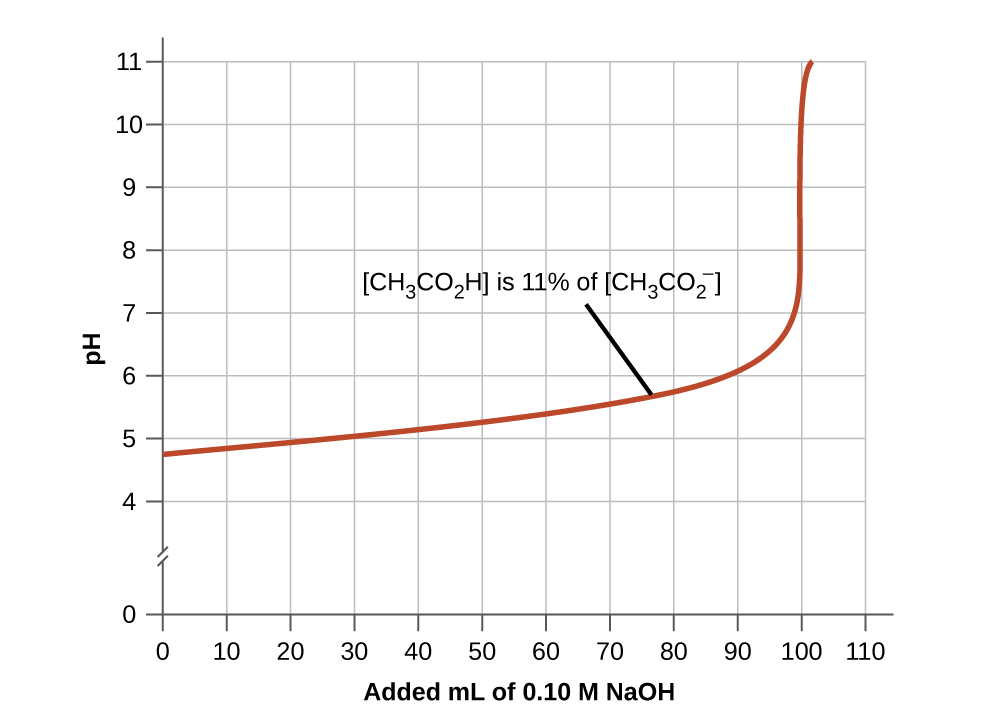
Figure 4. The graph, an illustration of buffering activeness, shows change of pH every bit an increasing amount of a 0.10-Thou NaOH solution is added to 100 mL of a buffer solution in which, initially, [CH3COtwoH] = 0.10 M and [CHthreeCOtwo −] = 0.10Grand. - Weak acids and their salts are better as buffers for pHs less than 7; weak bases and their salts are ameliorate as buffers for pHs greater than 7.
Blood is an of import case of a buffered solution, with the principal acid and ion responsible for the buffering activity being carbonic acid, H2COthree, and the bicarbonate ion, [latex]\text{HCO}_3^{\;\;-}[/latex]. When an excess of hydrogen ion enters the blood stream, it is removed primarily by the reaction:
[latex]\text{H}_3\text{O}^{+}(aq)\;+\;\text{HCO}_3^{\;\;-}(aq)\;{\longrightarrow}\;\text{H}_2\text{CO}_3(aq)\;+\;\text{H}_2\text{O}(fifty)[/latex]
When an excess of the hydroxide ion is present, it is removed by the reaction:
[latex]\text{OH}^{-}(aq)\;+\;\text{H}_2\text{CO}_3(aq)\;{\longrightarrow}\;\text{HCO}_3^{\;\;-}(aq)\;+\;\text{H}_2\text{O}(50)[/latex]
The pH of human being blood thus remains very near seven.35, that is, slightly basic. Variations are usually less than 0.ane of a pH unit. A change of 0.4 of a pH unit of measurement is probable to be fatal.
The Henderson-Hasselbalch Equation
The ionization-constant expression for a solution of a weak acid can be written as:
[latex]K_{\text{a}} = \frac{[\text{H}_3\text{O}^{+}][\text{A}^{-}]}{[\text{HA}]}[/latex]
Rearranging to solve for [H3O+], we get:
[latex][\text{H}_3\text{O}^{+}] = K_{\text{a}}\;\times\;\frac{[\text{HA}]}{[\text{A}^{-}]}[/latex]
Taking the negative logarithm of both sides of this equation, nosotros arrive at:
[latex]-\text{log}[\text{H}_3\text{O}^{+}] = -\text{log}\;K_{\text{a}}\;-\;\text{log}\frac{[\text{HA}]}{[\text{A}^{-}]}[/latex],
which can be written equally
[latex]\text{pH} = \text{p}K_{\text{a}}\;+\;\text{log}\frac{[\text{A}^{-}]}{[\text{HA}]}[/latex]
where pM a is the negative of the common logarithm of the ionization constant of the weak acid (pK a = −log Chiliad a). This equation relates the pH, the ionization abiding of a weak acrid, and the concentrations of the weak acrid and its table salt in a buffered solution. Scientists often employ this expression, called the Henderson-Hasselbalch equation, to calculate the pH of buffer solutions. It is important to note that the "x is small" assumption must exist valid to apply this equation.
Lawrence Joseph Henderson and Karl Albert Hasselbalch
Lawrence Joseph Henderson (1878–1942) was an American physician, biochemist and physiologist, to name only a few of his many pursuits. He obtained a medical degree from Harvard so spent 2 years studying in Strasbourg, so a part of Deutschland, earlier returning to take a lecturer position at Harvard. He eventually became a professor at Harvard and worked there his unabridged life. He discovered that the acid-base balance in human blood is regulated by a buffer system formed by the dissolved carbon dioxide in blood. He wrote an equation in 1908 to describe the carbonic acid-carbonate buffer system in blood. Henderson was broadly knowledgeable; in addition to his important inquiry on the physiology of blood, he also wrote on the adaptations of organisms and their fit with their environments, on folklore and on university education. He likewise founded the Fatigue Laboratory, at the Harvard Business Schoolhouse, which examined homo physiology with specific focus on piece of work in industry, practice, and diet.
In 1916, Karl Albert Hasselbalch (1874–1962), a Danish doc and chemist, shared authorship in a paper with Christian Bohr in 1904 that described the Bohr effect, which showed that the ability of hemoglobin in the claret to bind with oxygen was inversely related to the acerbity of the blood and the concentration of carbon dioxide. The pH scale was introduced in 1909 by another Dane, Sørensen, and in 1912, Hasselbalch published measurements of the pH of blood. In 1916, Hasselbalch expressed Henderson's equation in logarithmic terms, consistent with the logarithmic scale of pH, and thus the Henderson-Hasselbalch equation was born.
Medicine: The Buffer System in Blood
The normal pH of human blood is virtually 7.iv. The carbonate buffer system in the blood uses the following equilibrium reaction:
[latex]\text{CO}_2(k)\;+\;2\text{H}_2\text{O}(l)\;{\rightleftharpoons}\;\text{H}_2\text{CO}_3(aq)\;{\rightleftharpoons}\;\text{HCO}_3^{\;\;-}(aq)\;+\;\text{H}_3\text{O}^{+}(aq)[/latex]
The concentration of carbonic acrid, HtwoCO3 is approximately 0.0012 K, and the concentration of the hydrogen carbonate ion, [latex]\text{HCO}_3^{\;\;-}[/latex], is around 0.024 1000. Using the Henderson-Hasselbalch equation and the pG a of carbonic acid at body temperature, we can calculate the pH of blood:
[latex]\text{pH} = \text{p}K_{\text{a}}\;+\;\text{log}\frac{[\text{base of operations}]}{[\text{acid}]} = 6.iv\;+\;\text{log}\frac{0.024}{0.0012} = 7.7[/latex]
The fact that the H2CO3 concentration is significantly lower than that of the [latex]\text{HCO}_3^{\;\;-}[/latex] ion may seem unusual, only this imbalance is due to the fact that most of the by-products of our metabolism that enter our bloodstream are acidic. Therefore, at that place must be a larger proportion of base of operations than acrid, and then that the chapters of the buffer volition not be exceeded.
Lactic acrid is produced in our muscles when we exercise. As the lactic acid enters the bloodstream, it is neutralized by the [latex]\text{HCO}_3^{\;\;-}[/latex] ion, producing H2CO3. An enzyme so accelerates the breakdown of the excess carbonic acrid to carbon dioxide and water, which can exist eliminated by animate. In fact, in addition to the regulating furnishings of the carbonate buffering system on the pH of blood, the body uses breathing to regulate blood pH. If the pH of the claret decreases likewise far, an increase in breathing removes CO2 from the blood through the lungs driving the equilibrium reaction such that [HthreeO+] is lowered. If the claret is also element of group i, a lower jiff rate increases COii concentration in the blood, driving the equilibrium reaction the other manner, increasing [H+] and restoring an advisable pH.

View information on the buffer system encountered in natural waters.
Cardinal Concepts and Summary
A solution containing a mixture of an acrid and its conjugate base, or of a base of operations and its conjugate acrid, is called a buffer solution. Different in the case of an acid, base, or common salt solution, the hydronium ion concentration of a buffer solution does non change profoundly when a small-scale corporeality of acrid or base of operations is added to the buffer solution. The base (or acid) in the buffer reacts with the added acid (or base).
Primal Equations
- pThou a = −log K a
- pK b = −log Grand b
- [latex]\text{pH} = \text{p}K_{\text{a}}\;+\;\text{log}\frac{[\text{A}^{-}]}{[\text{HA}]}[/latex]
Chemical science Finish of Chapter Exercises
- Explain why a buffer can be prepared from a mixture of NH4Cl and NaOH only not from NHthree and NaOH.
- Explain why the pH does not modify significantly when a small amount of an acid or a base is added to a solution that contains equal amounts of the acid H3POiv and a salt of its conjugate base NaH2PO4.
- Explicate why the pH does not change significantly when a small amount of an acid or a base of operations is added to a solution that contains equal amounts of the base NH3 and a salt of its conjugate acid NH4Cl.
- What is [H3O+] in a solution of 0.25 M CH3CO2H and 0.030 M NaCH3CO2?
[latex]\text{CH}_3\text{CO}_2\text{H}(aq)\;+\;\text{H}_2\text{O}(l)\;{\rightleftharpoons}\;\text{H}_3\text{O}^{+}(aq)\;+\;\text{CH}_3\text{CO}_2^{\;\;-}(aq)\;\;\;\;\;\;\;K_{\text{a}} = one.viii\;\times\;10^{-v}[/latex] - What is [H3O+] in a solution of 0.075 One thousand HNOii and 0.030 M NaNOii?
[latex]\text{HNO}_2(aq)\;+\;\text{H}_2\text{O}(50)\;{\rightleftharpoons}\;\text{H}_3\text{O}^{+}(aq)\;+\;\text{NO}_2^{\;\;-}(aq)\;\;\;\;\;\;\;K_{\text{a}} = four.5\;\times\;10^{-v}[/latex] - What is [OH−] in a solution of 0.125 M CH3NHtwo and 0.130 M CH3NHiiiCl?
[latex]\text{CH}_3\text{NH}_2(aq)\;+\;\text{H}_2\text{O}(50)\;{\rightleftharpoons}\;\text{CH}_3\text{NH}_3^{\;\;+}(aq)\;+\;\text{OH}^{-}(aq)\;\;\;\;\;\;\;K_{\text{b}} = 4.4\;\times\;10^{-iv}[/latex] - What is [OH−] in a solution of 1.25 M NH3 and 0.78 M NHfourNO3?
[latex]\text{NH}_3(aq)\;+\;\text{H}_2\text{O}(l)\;{\rightleftharpoons}\;\text{NH}_4^{\;\;+}(aq)\;+\;\text{OH}^{-}(aq)\;\;\;\;\;\;\;K_{\text{b}} = 1.8\;\times\;10^{-5}[/latex] - What concentration of NH4NO3 is required to make [OH−] = 1.0 × 10−5 in a 0.200-M solution of NHthree?
- What concentration of NaF is required to make [H3O+] = ii.3 × ten−iv in a 0.300-M solution of HF?
- What is the consequence on the concentration of acetic acid, hydronium ion, and acetate ion when the following are added to an acidic buffer solution of equal concentrations of acerb acid and sodium acetate:
(a) HCl
(b) KCH3CO2
(c) NaCl
(d) KOH
(east) CHthreeCO2H
- What is the effect on the concentration of ammonia, hydroxide ion, and ammonium ion when the following are added to a basic buffer solution of equal concentrations of ammonia and ammonium nitrate:
(a) KI
(b) NHthree
(c) Howdy
(d) NaOH
(e) NH4Cl
- What will exist the pH of a buffer solution prepared from 0.20 mol NH3, 0.40 mol NH4NO3, and but enough water to give 1.00 L of solution?
- Calculate the pH of a buffer solution prepared from 0.155 mol of phosphoric acid, 0.250 mole of KH2PO4, and enough water to make 0.500 L of solution.
- How much solid NaCHthreeCO2•3H2O must be added to 0.300 L of a 0.50-Grand acerb acid solution to requite a buffer with a pH of 5.00? (Hint: Assume a negligible change in volume every bit the solid is added.)
- What mass of NH4Cl must be added to 0.750 Fifty of a 0.100-M solution of NHiii to give a buffer solution with a pH of 9.26? (Hint: Assume a negligible change in volume as the solid is added.)
- A buffer solution is prepared from equal volumes of 0.200 M acerb acid and 0.600 Yard sodium acetate. Utilize 1.fourscore × 10−five as K a for acetic acid.
(a) What is the pH of the solution?
(b) Is the solution acidic or basic?
(c) What is the pH of a solution that results when 3.00 mL of 0.034 M HCl is added to 0.200 L of the original buffer?
- A 5.36–g sample of NHfourCl was added to 25.0 mL of 1.00 Thou NaOH and the resulting solution
diluted to 0.100 50.
(a) What is the pH of this buffer solution?
(b) Is the solution acidic or basic?
(c) What is the pH of a solution that results when 3.00 mL of 0.034 M HCl is added to the solution?
- Which acid in Tabular array ii in Chapter 14.3 Relative Strengths of Acids and Bases is virtually appropriate for grooming of a buffer solution with a pH of three.1? Explain your choice.
- Which acrid in Tabular array two in Chapter xiv.3 Relative Strengths of Acids and Bases is most appropriate for preparation of a buffer solution with a pH of 3.seven? Explain your choice.
- Which base in Table 3 in Affiliate fourteen.3 Relative Strengths of Acids and Bases is almost appropriate for preparation of a buffer solution with a pH of 10.65? Explain your choice.
- Which base of operations in Table 3 in Chapter 14.3 Relative Strengths of Acids and Bases is most appropriate for preparation of a buffer solution with a pH of 9.20? Explain your choice.
- What is the pH of 1.000 L of a solution of 100.0 g of glutamic acrid (C5HnineNOiv, a diprotic acid; K 1 = 8.5 × 10−5, K ii = 3.39 × 10−ten) to which has been added 20.0 chiliad of NaOH during the training of monosodium glutamate, the flavoring agent? What is the pH when exactly 1 mol of NaOH per mole of acid has been added?
Glossary
- buffer capacity
- amount of an acid or base that can be added to a volume of a buffer solution before its pH changes significantly (usually past one pH unit)
- buffer
- mixture of a weak acrid or a weak base and the salt of its conjugate; the pH of a buffer resists change when modest amounts of acid or base are added
- Henderson-Hasselbalch equation
- equation used to calculate the pH of buffer solutions
Solutions
Answers to Chemistry End of Chapter Exercises
two. Backlog H3O+ is removed primarily by the reaction:
[latex]\text{H}_3\text{O}^{+}(aq)\;+\;\text{H}_2\text{PO}_4^{\;\;-}(aq)\;{\longrightarrow}\;\text{H}_3\text{PO}_4(aq)\;+\;\text{H}_2\text{O}(l)[/latex]
Excess base of operations is removed past the reaction:
[latex]\text{OH}^{-}(aq)\;+\;\text{H}_3\text{PO}_4(aq)\;{\longrightarrow}\;\text{H}_2\text{PO}_4^{\;\;-}(aq)\;+\;\text{H}_2\text{O}(50)[/latex]
4. [HiiiO+] = i.5 × 10−4 G
6. [OH−] = 4.2 × x−4 M
8. [NH4NO3] = 0.36 Grand
10. (a) The added HCl will increment the concentration of HiiiO+ slightly, which will react with [latex]\text{CH}_3\text{CO}_2^{\;\;-}[/latex] and produce CH3CO2H in the procedure. Thus, [latex][\text{CH}_3\text{CO}_2^{\;\;-}][/latex] decreases and [CH3CO2H] increases.
(b) The added KCH3CO2 volition increase the concentration of [latex][\text{CH}_3\text{CO}_2^{\;\;-}][/latex] which will react with H3O+ and produce CHiiiCOii H in the process. Thus, [HiiiO+] decreases slightly and [CH3COtwoH] increases.
(c) The added NaCl will have no effect on the concentration of the ions.
(d) The added KOH will produce OH− ions, which will react with the H3O+, thus reducing [H3O+]. Some additional CH3COiiH will dissociate, producing [latex][\text{CH}_3\text{CO}_2^{\;\;-}][/latex] ions in the procedure. Thus, [CH3COtwoH] decreases slightly and [latex][\text{CH}_3\text{CO}_2^{\;\;-}][/latex] increases.
(e) The added CH3COiiH will increase its concentration, causing more of it to dissociate and producing more [latex][\text{CH}_3\text{CO}_2^{\;\;-}][/latex] and H3O+ in the process. Thus, [H3O+] increases slightly and [latex][\text{CH}_3\text{CO}_2^{\;\;-}][/latex] increases.
12. pH = 8.95
xiv. 37 g (0.27 mol)
16. (a) pH = 5.222;
(b) The solution is acidic.
(c) pH = 5.221
eighteen. To prepare the best buffer for a weak acid HA and its salt, the ratio [latex]\frac{[\text{H}_3\text{O}^{+}]}{K_{\text{a}}}[/latex] should be equally close to i as possible for constructive buffer action. The [H3O+] concentration in a buffer of pH 3.1 is [H3O+] = 10−3.1 = seven.94 × 10−four M
Nosotros can at present solve for Thou a of the best acid as follows:
[latex]\frac{[\text{H}_3\text{O}^{+}]}{K_{\text{a}}} = 1 \\[0.5em] K_{\text{a}} = \frac{[\text{H}_3\text{O}^{+}]}{1} = seven.94\;\times\;ten^{-four}[/latex]
In Table 2 in Affiliate 14.3 Relative Strengths of Acids and Bases, the acid with the closest Chiliad a to 7.94 × x−iv is HF, with a K a of 7.2 × ten−4.
20. For buffers with pHs > 7, yous should use a weak base and its salt. The most effective buffer will take a ratio [latex]\frac{[\text{OH}^{-}]}{K_{\text{b}}}[/latex] that is as close to one equally possible. The pOH of the buffer is 14.00 − 10.65 = 3.35. Therefore, [OH−] is [OH−] = x−pOH = 10−three.35 = 4.467 × 10−4 One thousand.
We tin now solve for Grand b of the best base as follows:
[latex]\frac{[\text{OH}^{-}]}{K_{\text{b}}} = i[/latex]
K b = [OH−] = 4.47 × 10−4
In Tabular array three in Chapter 14.3 Relative Strengths of Acids and Bases, the base of operations with the closest One thousand b to 4.47 × 10−4 is CH3NHtwo, with a 1000 b = four.four × ten−four.
Source: https://opentextbc.ca/chemistry/chapter/14-6-buffers/
0 Response to "What Is the Ph When 1.5 Ml of 3.0 M Hcl Is Added to 1.0 L of the Buffer?"
Post a Comment